GE Healthcare today announced an agreement to invest up to $50 million in Israeli start-up Pulsenmore. The investment is aimed at accelerating global adoption of Pulsenmore’s homecare ultrasound solutions and will also support the company's goal to pursue U.S. FDA clearance and commercial expansion.
In addition to its equity investment, GE Healthcare will also partner with Pulsenmore to distribute its products in Europe and other markets as the product becomes available for commercial distribution and collaborate closely on developing ultrasound-based healthcare offerings that cater to the burgeoning homecare market, which is expected to reach USD $662.67 billion by 2027 and poised to grow at a compound annual growth rate of 14.2%[i].
The telehealth market is projected to expand at a compound annual growth rate (CAGR) of over 30% from 2022-2029[ii], creating a need for robust technologies that can improve remote monitoring and clinical decision-making.
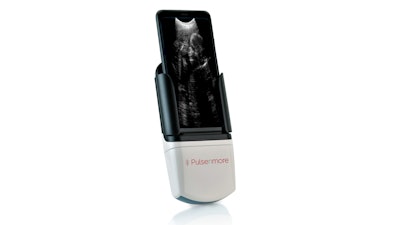
Pulsenmore is well positioned for this market with its novel self-operated prenatal home ultrasound solution, which, combined with a smartphone, enables pregnant women to self-scan for remote clinical assessment by their healthcare provider.
The complete product offering includes a handheld ultrasound device that docks with the user’s smartphone; a mobile app for the patient to conduct offline/online consultation with a clinician; a web application for clinician-side interaction with the platform; and a software API enabling integration of Pulsenmore’s online services with organization health records. The periodic fetal ultrasound scanning and consultation enabled by the Pulsenmore device can be offered as an online clinician-guided telecommunication service or as an offline application-guided service.
 Pulsenmore
Pulsenmore
Additional applications and devices are currently under development, including remote follicular monitoring for women undergoing in-vitro fertilization (IVF) as well as remote monitoring for chronic heart failure (CHF) and end-stage renal disease (ESRD).
The Pulsenmore prenatal ultrasound platform has already been implemented successfully across Israel in partnership with the country’s largest HMO, Clalit Health Services. Most recently, physicians at Israel’s Sheba Medical Center have been using the device to conduct prenatal ultrasounds on refugees at the Ukraine border as part of a new 'virtual hospital' that has Israeli medical staff caring for people injured or displaced in the conflict.
 Pulsenmore
Pulsenmore
[i] Precedence research – Global Home Healthcare Market 2016-2027
[ii] Grand View Research – Telehealth Market Size & Share Report 2022-2028 – “CAGR of 36.8% from 2022-2029”






















