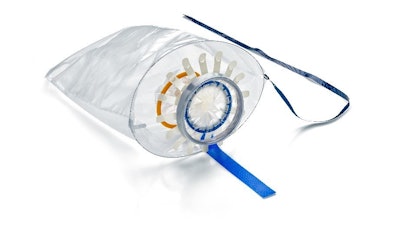
Olympus announced today the market launch of the Guardenia Contained Extraction System. Guardenia is the latest innovation in their contained tissue extraction product portfolio for gynecological procedures and is manufactured by Advanced Surgical Concepts (ASC) Ltd. of Bray, Ireland.
Olympus America is the exclusive U.S. distributor of the system, which is indicated to contain and isolate tissue during, or prior to, surgical removal and/or extracorporeal manual morcellation.
The Guardenia system is designed to prevent the escape of cells into the abdominal cavity during extracorporeal manual morcellation, and its integrated guard provides protection against inadvertent damage from sharp surgical instruments around the incision. The guard offers superior incision retraction and protects a greater area of the containment element when compared to currently marketed devices.1
Guardenia maximizes the amount of available space in the incision during a procedure by encroaching less into the incision and maintaining a stable protective area to complete manual tissue morcellation. Once in place, the design of the containment bag and guard ensures more durable protection of the specimen bag and the incision as compared to other marketed devices.
Protection of the integrity of the specimen bag is one of the critical design features of the Guardenia device. Guardenia's design securely anchors the guard and prevents bag dislodgement.
Other features of Guardenia include:
- Can be deployed through a standard 12mm trocar, under pneumoperitoneum and with or without vision;
- Guardenia is universal, and one size fits all;
- The guard automatically conforms to the shape and size of the incision to provide maximal protection of the containment bag;
- The guard is securely anchored in the incision and cannot become dislodged during tissue extraction;
- Employs "Set and forget" technology, allowing the surgeon to focus on the process of morcellation;
- Superior retraction of the incision, maximizing the working area for more ergonomic morcellation;
- Maximizes the ratio of incision size to extraction opening;
- Bag material is designed to be impermeable to the passage of liquid or tissue, measured down to the cellular level;
- The device works across multiport, robotic and single-port surgery;
- No additional training is necessary.
Potential complications associated with the use of the Guardenia device are the same as those associated with the use of tissue or specimen bags, morcellation and laparoscopic surgery in general and include but are not limited to superficial lesions, injury to internal vessels, bleeding, hematoma, injury to the abdominal wall, injury to bowel, infection, peritonitis and spread of benign or malignant tissue.






















