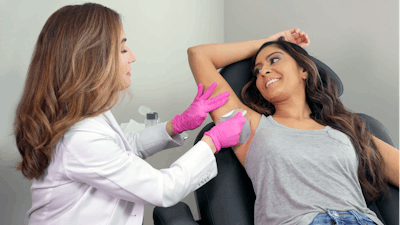
Candesant Biomedical said the FDA has granted a de novo request for Brella, a "3-Minute SweatControl Patch" for the reduction of excessive underarm sweating in adults with primary axillary hyperhidrosis. Brella uses Candesant's novel, patented TAT (targeted alkali thermolysis) technology. The Brella 3-Minute SweatControl Patch provides a new in-office approach to sweat control that is fast, non-invasive, needle-free, aluminum-free, and affordable, with results lasting three to four months.
"FDA clearance of Brella represents a significant advance for millions of people dealing with the physical, emotional, financial, and aesthetic impact of hyperhidrosis, or sweating beyond what's needed to keep the body cool," said Niquette Hunt, Founder and CEO of Candesant. "Satisfaction with current treatments is low3 and 80% of consumers are seeking new treatments to manage excessive underarm sweat.4 Brella provides a new option for a common condition that curtails activities, stains clothes, causes frustration and embarrassment, undermines confidence, and has been inadequately treated for far too long."
Consisting of a sodium sheet with an adhesive overlay, the Brella SweatControl Patch is a single-use disposable patch applied to a patient's underarm by a healthcare provider for up to three minutes. Its mechanism of action utilizes Candesant's patented TAT technology, which is based on the scientific principle that heat is generated when sodium comes in contact with water in sweat. The thermal energy created by the sodium sheet is precisely localized, microtargeting sweat glands to significantly reduce sweat production. The Brella SweatControl Patch is the first clinical application to harness and target this well-established property of sodium and is the only patented medical device based on it.
Brella will be available in select U.S. markets beginning late summer of 2023 through the Candesant Brella Early Experience Program, which will include healthcare providers with aesthetic practices focused on providing their patients with the latest innovations in sweat control and will be launched nationally following the Brella Early Experience Program.





















