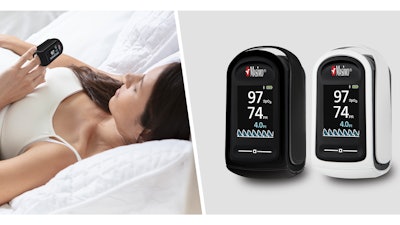
Masimo announced today FDA clearance of MightySat Medical, which the company said makes it the first and only FDA-cleared medical fingertip pulse oximeter available over the counter (OTC) direct to consumers without a prescription.
This clearance brings consumers a pulse oximeter medical device powered by Masimo SET pulse oximetry. Starting today, the MightySat Medical Pulse Oximeter can be purchased at masimo.com, and it will soon be available for purchase at retail and drug stores across the country.
In 2021, the FDA issued a safety communication clarifying that it does not review the accuracy of “health and wellness” pulse oximeters and highlighting the common limitations of both health and wellness and some prescription pulse oximeters—such as difficulty measuring accurately during patient motion, on patients with poor circulation (low perfusion), and on darker skin pigmentation. The overwhelming majority of consumers say that device accuracy—measurements they can trust—is their top priority when choosing a pulse oximeter. MightySat Medical provides a direct-to-consumer option for a medical device with hospital-grade pulse oximetry technology, Masimo SET, whose performance has been reviewed and cleared by the FDA for medical use. MightySat Medical provides accurate and reliable oxygen saturation (SpO2) and pulse rate (PR) measurements on patients of all skin colors and patients with low perfusion.






















