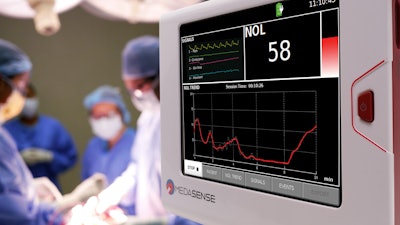
Medasense Biometrics announced today that the U.S. Food and Drug Administration (FDA) has granted marketing authorization to the PMD-200 patient monitor with NOL technology developed for monitoring nociception (the physiological response to pain) through the De Novo premarket review pathway.
NOL uses a unique multi-parametric sensor platform and advanced AI algorithms to convert complicated data into a patient's ‘Signature of Pain.’ The technology, which is currently utilized in operating rooms and high-acuity settings, where patients are under anesthesia and unable to communicate, enables clinicians to personalize pain management, control pain and avoid overmedication. NOL is the first and only market-authorized, adjunctive monitoring technology for the assessment of changes in nociception in adult patients receiving opioid or opioid sparing analgesics.
“NOL technology has the potential to improve the lives of hundreds of millions of patients worldwide, and we have already seen the impact it has had in multiple countries,” said Galit Zuckerman-Stark, CEO & founder of Medasense. “Our mission is to help patients suffer less from pain and the adverse effects of pain medication. We are proud to offer the first and only measure of intraoperative pain (nociception) in the US, and to be able to offer meaningful innovation in the area of pain management to help patients undergoing surgery experience improved pain-related outcomes.”
Studies have shown that intraoperative NOL monitoring can reduce postoperative pain experienced by patients in the post anesthesia care unit, and potentially reduce costs of care. A recent publication showed that the odds of suffering severe post-operative pain are six times lower with NOL monitoring. This addresses a widespread need. It is estimated that 50% of surgical patients suffer from moderate to severe postoperative pain and 12% suffer adverse events due to pain relief medication. These can result in extended hospitalization, additional healthcare costs, and a 50% increase in hospital readmissions.
“The anesthesia community has needed a technology like NOL for a long time,” said Frank Overdyk, MSEE, MD, Charleston, SC. “We have devices that monitor depth of anesthesia, we have TOF cuffs to check for patient movement, but the missing piece of the puzzle is a way to monitor the effect of the opioid or opioid sparing analgesia. Relying on patient’s heart rate and blood pressure is neither specific nor sensitive enough to pain. This technology as an adjunctive to clinical judgment will provide a window into the patient’s nociceptive state during surgery so we can personalize the way we administer analgesia, improving the patient’s recovery.”