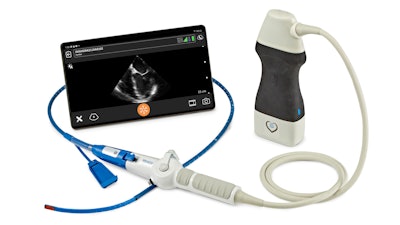
Clarius Mobile Health, a provider of high-definition wireless ultrasound systems, and ImaCor, a hemodynamic ultrasound company, today announced a partnership that makes available a handheld transesophageal echocardiography (TEE) system.
The Zura Handheld Hemodynamic Ultrasound system powered by Clarius provides an instant, clear window to directly visualize preload and contractility over time, having received clearance by the U.S. Food and Drug Administration.
"A high-risk patient's hemodynamic status changes dramatically in a matter of minutes, often while clinicians are waiting for test results they submitted 15 minutes ago," said ImaCor Founder Scott Roth, MD. "The Zura Handheld provides instant and accurate information to guide patient management and it's proven to save lives. More than 20,000 patients have already benefitted from our cart-based Zura system. We're excited to make this life-saving device now highly accessible to more high acuity care teams with the introduction of our handheld system, which is easier to bring to the bedside, can be used to monitor multiple patients in real-time, and takes up no floor space. We chose to partner with Clarius for our handheld solution, because it delivers the highest image quality for the best patient care."
The Zura Handheld Hemodynamic Ultrasound system is the companion device to the ClariTEE transesophageal echo probe. Now powered by Clarius, they comprise an advanced handheld hemodynamic management platform for real-time decision making to care for the most at-risk patients. The 22-ounce handheld is a key advantage in the ICU where space is limited. The Zura Handheld operates with the Clarius Ultrasound App, which connects wirelessly to Apple and Android smart devices.