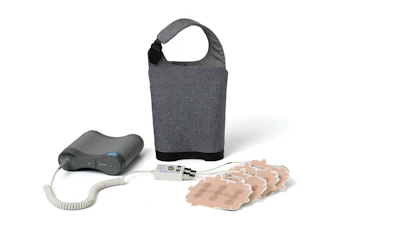
Novocure said the FDA has approved Optune Lua for concurrent use with PD-1/PD-L1 inhibitors or docetaxel, for the treatment of adult patients with metastatic non-small cell lung cancer (mNSCLC) who have progressed on or after a platinum-based regimen.
Optune Lua is a portable device that produces alternating electric fields known as tumor treating fields (TTFields), which are delivered through non-invasive, wearable arrays. TTFields exert physical forces on the electrically charged components of dividing cancer cells, resulting in cell death.
“There have been a number of important advances in first-line treatment for NSCLC, but this is an aggressive disease, and most patients will develop progression, with limited effective treatment options in second line and beyond,” said Ticiana Leal, MD, Associate Professor and Director of the Thoracic Oncology Program at the Winship Cancer Institute of Emory University School of Medicine and primary investigator of the LUNAR study. “The overall survival results we observed with Optune Lua in the LUNAR study mark the first substantial improvement in more than 8 years in this patient population which, when combined with Optune Lua’s lack of systemic toxicity, make this a compelling development for many patients and their physicians who need better treatment options for this advanced disease.”
“We are excited patients with metastatic NSCLC have more options, which they urgently need,” said GO2 for Lung Cancer Chief Patient Officer Danielle Hicks. “The fight against lung cancer is always evolving, and the number of people affected by this disease is underappreciated. That is why Novocure’s commitment to advancing treatment is exciting for the whole lung cancer community.”
Lung cancer is the one of the most common causes of cancer-related death worldwide, and non-small cell lung cancer (NSCLC) accounts for approximately 85% of all lung cancers. It is estimated that approximately 193,000 patients are diagnosed with NSCLC each year in the U.S.
Physicians use different combinations of surgery, radiation and pharmacological therapies to treat NSCLC, depending on the stage of the disease. Surgery, which may be curative in a subset of patients, is usually used in early stages of the disease. Since 1991, radiation with a combination of platinum-based chemotherapy drugs has been the first-line standard of care for locally advanced or metastatic NSCLC. Certain immune checkpoint inhibitors, including both PD-1 and PD-L1 inhibitors, have been approved for the first-line treatment of NSCLC and the standard of care in this setting continues to evolve rapidly.
It is estimated that approximately 30,000 patients actively seek treatment for stage 4 NSCLC after progressing during or after platinum-based therapy each year in the U.S. The standard of care for second-line treatment is also evolving and may include platinum-based chemotherapy for patients who received immune checkpoint inhibitors as their first-line regimen, pemetrexed, docetaxel or immune checkpoint inhibitors.