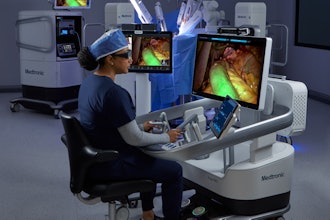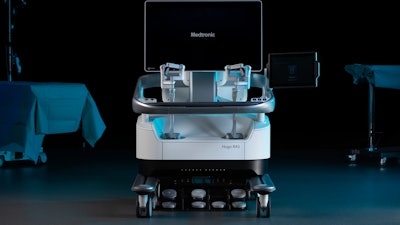
Medtronic today announced the first patient enrolled in the Expand URO U.S. clinical trial for the Hugo robotic-assisted surgery (RAS) system. The robotic-assisted prostatectomy procedure was performed by Dr. Michael R. Abern at Duke University Hospital in Durham, N.C.
Minimally invasive surgery, including robotic-assisted surgery, offers fewer complications, shorter hospital stays, faster return to normal activities, and smaller scars. Urologic procedures are one of the most commonly performed with the assistance of a surgical robot. The Hugo RAS system is intended to be used in this study for urologic surgical procedures including radical prostatectomy, radical cystectomy, and nephrectomy (partial or radical) procedures at sites in the U.S.
"Scientific evidence is the bedrock of healthcare technology innovation and adoption. It creates and builds trust among clinicians and patients," said Carla Peron, M.D., chief medical officer of the Surgical Robotics business, which is part of the Medical Surgical Portfolio at Medtronic. "We're proud to further that important pursuit with the Hugo RAS system in partnership with hospitals and surgical teams in the United States who share our commitment to patients."
The Expand URO clinical trial is being conducted pursuant to an Investigational Device Exemption from the U.S. Food and Drug Administration (FDA). Up to 122 patients will be enrolled in the study at six sites in the U.S.
Dr. James Porter, a urologic surgeon at Swedish Medical Center in Seattle, WA, is the principal investigator of the U.S. Expand URO study.
"This is an exciting time for healthcare in the United States and around the world, as we have the opportunity to expand robotic-assisted surgery treatment options to more patients," said Dr. Porter, who plans to perform his first cases under the trial this month. "Backed by a growing body of clinical evidence, robotic-assisted surgery is the preferred approach within urology given the anatomical access, precision, and ergonomic advantages it enables."
The Hugo RAS system, combined with Touch Surgery Enterprise, offers a smart, digitally enabled surgical experience. Outside the U.S., it is in use at hospitals across three continents in a range of procedures within urology, gynecology, and general surgery.
The Hugo RAS system is commercially available in certain geographies. Regulatory requirements and status in individual countries and regions will determine market availability of the Hugo RAS system and approved indications. In the U.S., the Hugo RAS system is an investigational device not for sale.