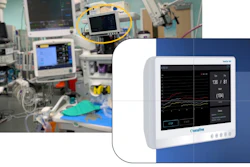
The Intera 3000 Hepatic Artery Infusion Pump is used to continuously deliver medicine through an artery that supplies blood and oxygen to the liver and several other organs (hepatic artery).
The pump is implanted into adult patients and delivers chemotherapy medications to treat liver tumors.
People with the Intera 3000 Hepatic Artery Infusion Pump are able to get back to daily activities and return to the hospital for pump refills every two weeks.
Intera Oncology is recalling the device after receiving reports from clinicians that the pumps were delivering medications (flow rate) faster than expected.
If the pump delivers infusions at faster than expected flow rates, the patient may receive too much medication at one time, resulting in life-threatening hematologic (myelosuppression) toxicity, neurotoxicity or death.
Additionally, if the flow rate is too fast, the patient may run out of medication before a pump refill occurs, which may allow their disease to progress or lead to death.
There have been three incidents reported related to this issue, with no injuries or deaths.
On July 10, 2022, Intera Oncology requested health care personnel and patients perform the following actions:
- Continue to monitor flow rate per standard refill procedure.
- Continue to monitor for liver toxicity to adjust dosing as per standard protocols.
- Consider pump replacement if altered flow rate of the pump can’t be adequately managed by dosing adjustments of the infusate when hepatic artery infusion therapy needs to continue.
- Consider bringing patient in for medication refill and to verify the flow rate sooner than every two weeks if the pump appears to be flowing more than 15% outside of labeled specification.
- Flush the bolus pathway with low dose heparinized saline (100 IU/ml) using the special bolus needle (per the Instructions for Use) if the pump is empty at the time of refill.
- The hepatic artery infusion therapy care team should decide whether or not to inform the patient about the possibility of alterations in flow.
- Notify any new clinicians about the possibility of altered flow in the future if a patient’s care is transferred to a new treating facility.
For more information, contact Intera Oncology at (800) 660-2660 or [email protected].