3M Health Care today announced the introduction of its new FDA-approved 3M SoluPrep S Sterile Antiseptic Solution chlorhexidine gluconate (2% w/v) and isopropyl alcohol (70% v/v) Patient Preoperative Skin Preparation. The product has been shown to provide fast-acting, broad-spectrum antimicrobial activity (in vitro, clinical significance unknown) and persistence in healthy human volunteers for at least 96-hours post-prep.
3M SoluPrep S Sterile Antiseptic Solution's optimized features and functionalities include:
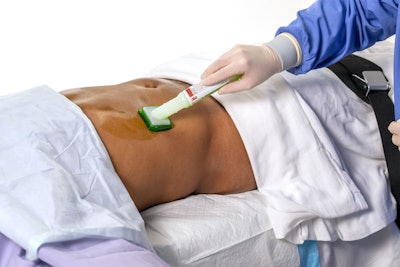
- Brite Green tint, which can be seen on a variety of skin tones, to help users ensure the surgical prep area is covered completely.
- Designed with work flow efficiency and convenience in mind, the applicator provides greater coverage area compared to the leading CHG/IPA surgical prep product with the same size applicator.
- Applicator activation is easy and quick, and the foam sponge controls the flow of solution during application to minimize drip.
- Contains an ingredient that can improve incise drape adherence.
A limited commercial release of 3M SoluPrep S Sterile Antiseptic Solution will begin in the U.S. on April 3, 2023. The product's full commercial launch will take place in 2024.
The 3M Medical Solutions Division skin and wound care portfolio includes a comprehensive range of integrated surgical and skin care solutions -- advanced and acute wound care dressings and products, medical tapes, sterilization products, patient prep and warming products, advanced wound care and specialty surgical applications, etc. – across the care continuum, from products that protect and maintain the integrity of skin to a full portfolio of advanced surgical wound care solutions.