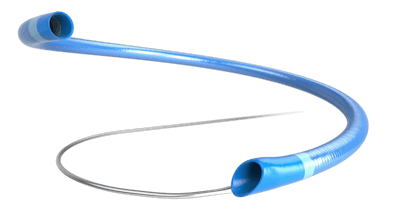
MIVI Neuroscience today announced the first patient enrollments in a multicenter clinical study to assess the safety and performance of the Q Aspiration Catheter used in a combined aspiration with stent-retriever technique for thrombus removal in patients who have suffered an acute ischemic stroke.
The AQUAS Registry is a prospective registry of 250 subjects conducted across up to 12 high-volume, comprehensive stroke centers in Europe. It is the largest study of a MIVI device to date. The study is expected to conclude within 12 months.
In the United States, approximately 50% of ischemic stroke interventions employ a combined technique; the other 50% use only an aspiration catheter.
Led by Dr. Sebastian Remollo Friedemann of Hospital Universitari Germans Trias I Pujol in Barcelona, the registry will assess the safety and performance of the Q Aspiration Catheter combined with the Acandis Aperio stent-retriever in treating ischemic stroke patients.
The Q Aspiration Catheter is CE-marked in Europe and under Investigational Device Exemption in the U.S.