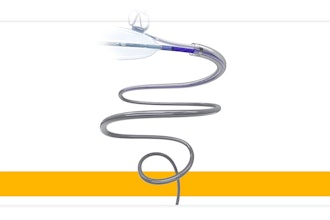
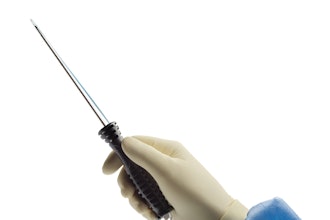
Agilis Robotics said it has made technological breakthroughs in developing highly miniaturized and flexible robotic instruments that offer dexterity even in the most challenging anatomies. This achievement empowers surgeons to perform intricate tissue resection procedures with precision and ease, directly within natural orifices. The system integrates with standard endoscopes used by hospitals and clinics, including colonoscopes and cystoscopes, leveraging existing endoscopic expertise, flattening the learning curve, and improving cost-effectiveness. The robotic instruments offer possibilities across endoluminal surgery, spanning the upper and lower gastrointestinal (GI) tract, urinary tract, throat, and gynecology.
The system consists of a primary cart, a compact control console, and disposable robotic arms which ensure performance and sterility. The cost of the system will be less than 20% of conventional robotic systems on the market, resulting in lower upfront investment and higher gross profits for medical institutions. The demand for endoluminal surgeries is surging worldwide, driven by increasing cancer screening and detection of early-stage tumours. With more than 3.5 million new GI cancer cases and 690k bladder cancer cases per year globally, Agilis Robotics is strategically positioned to capture the underserved endoluminal surgery market, allowing patients to receive effective early-stage treatment with reduced procedure time and recurrence rates.
Agilis Robotics is a spin-off from The University of Hong Kong and has secured more than HKD 90 million in funding from private investors. The funds are being utilized for further development and preparation for clinical trials. The company has completed six live animal studies with their clinical partners in Hong Kong and Shenzhen, demonstrating promising results for the system's accuracy, safety, and efficacy. First-in-human trials are targeted for the first half of 2024. The company is actively preparing for Food and Drug Administration (FDA) and National Medical Products Administration (NMPA) regulatory submissions, which mark a significant milestone towards commercialization.
The company's CFO, Hui Ka-Ming, with extensive experience in investing and pre-profit biotech companies, highlights the success of "Chapter 18A" listed companies in the HKEX, despite their relative immaturity compared to Agilis Robotics. Furthermore, Agilis Robotics understands the importance of cost-effectiveness, marketability, and market potential, ensuring a successful venture for investors and hospitals.