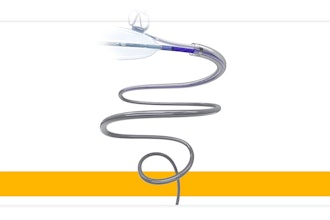
MMI (Medical Microinstruments, Inc.) today announced that its Symani Surgical System is now commercially available in the United States. The U.S. Food and Drug Administration (FDA) granted De Novo Classification to the robotic system for soft tissue manipulation to perform microsurgery, a highly specialized technique that involves reconnecting tiny vessels to restore blood flow or redirect fluid during reconstruction or repair.
The FDA authorization makes the Symani Surgical System the only commercially available platform in the U.S. for reconstructive microsurgery. The technology is positioned to open the field of microsurgery to new surgeons by quickly developing their skills, as well as to empower skilled microsurgeons to confidently expand into supermicrosurgery, creating a novel category of treatments that the human hand cannot perform without robotic assistance.
Specific techniques used in microsurgery include reconnecting small anatomical structures, such as blood and lymphatic vessels, during open surgical procedures. Supermicrosurgery involves reconstruction or repair of even smaller vessels, typically less than 1mm in diameter, and fewer than 600 surgeons worldwide perform supermicrosurgery today.
The Symani Surgical System provides advanced solutions for a range of open surgeries, including post-mastectomy breast cancer reconstruction, extremity reconstruction using free tissue transfer, and lymphatic system repair.
The Symani Surgical System offers surgeons entirely new capabilities because it features the world’s smallest surgical robotic wrist, called NanoWrist®. The unique design enables surgeons to replicate the natural movements of the human hand at the micro scale, which encourages a flatter learning curve in the training process. The articulated wrist features seven degrees of freedom that match the human wrist, tremor filtration and motion scaling, ultimately increasing precision and control.
Surgeons have leveraged the Symani Surgical System in nearly 1,000 clinical cases in the European Union and in thousands of preclinical cases around the world. The Symani Surgical System is available for commercial use in Europe and parts of Asia Pacific. MMI plans to immediately launch the technology in the U.S.