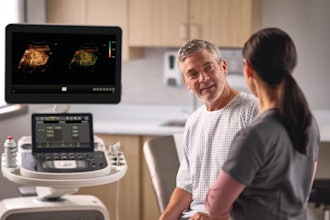
Boston Scientific today announced the close of its acquisition of Relievant Medsystems Inc., a company that offers the only U.S. Food and Drug Administration-cleared Intracept Intraosseous Nerve Ablation System, a therapy to treat vertebrogenic pain that is a form of chronic low back pain. The Intracept system, a basivertebral nerve ablation therapy, will be an addition to the Boston Scientific chronic pain portfolio that includes spinal cord stimulation, radiofrequency ablation and an interspinous spacer procedure.
Relievant recently secured national coverage from Anthem Blue Cross Blue Shield and Humana that are both effective now, expanding access to the Intracept system. These national payers join Cigna Healthcare and local Medicare plans enabling more access to treatments for people living with vertebrogenic pain. These policies expand access to more than 150 million lives.
"The revolutionary therapy from Relievant has tremendous potential to help even more people living with chronic pain," said Jim Cassidy, president, Neuromodulation, Boston Scientific. "The completion of this acquisition further differentiates our position in advanced interventional chronic pain, offering physicians the broadest selection of evidence-based treatment options that address multiple pain targets and change patients' lives."
The acquisition includes an upfront cash payment of $850 million and undisclosed additional contingent payments based on sales performance over the next three years. On an adjusted basis, the transaction is expected to be immaterial to adjusted earnings per share in 2024, slightly accretive in 2025 and increasingly accretive thereafter. On a GAAP basis, the transaction is expected to be more dilutive due to amortization expense and acquisition-related charges.