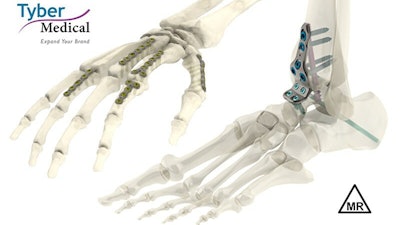
Tyber Medical, the global leader in private-label medical devices for major orthopedic trauma, extremity and spine companies, has been granted a 510(k) clearance by the U.S. Food and Drug Administration to expand its anatomical plate portfolio and an additional clearance for MR Conditional labeling. The company's latest FDA clearance extends the breath of its Anatomical Plating System with 391 additional plate configurations, including 100 new plates targeting foot, ankle and wrist indications.
Tyber Medical's 510(k) cleared plating system is indicated to treat a range of deformity, trauma, and degenerative conditions of the wrist, foot, ankle and long bones. The MR Conditional term allows Tyber Medical's Anatomical Plating System to be safely used in the MRI environment under certain tested conditions.
According to Tyber Medical's Director of Regulatory Affairs Lisa Boyle, the company has vigorously worked to meet the requirements necessary to gain this important designation from the FDA. "Over the past two years, the FDA has raised the bar for the industry to meet the MR compatibility requirements," said Boyle. "Our vertically integrated team has gone above and beyond to meet these requirements, and is very excited that our Anatomical Plating System has demonstrated safety in the MR environment, permitting us to label our system as MR Conditional."
Tyber Medical officially launched its Anatomical Plating System in 2020, when the product line received its initial 510(k) clearance from the FDA. Its latest FDA filing included new plate sizes, instrumentation and configurations for short bones, long bones and ankle fracture/fusion. This new submission also included ASTM testing and analysis, demonstrating the system can safely be used in the MRI environment.