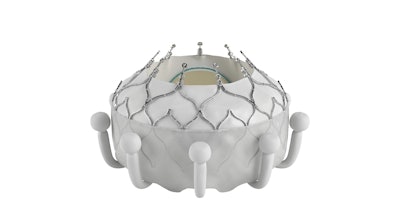
Edwards Lifesciences today announced the company’s EVOQUE tricuspid valve replacement system is the first transcatheter therapy to receive FDA approval for the treatment of tricuspid regurgitation (TR). The EVOQUE system is indicated for the improvement of health status in patients with symptomatic severe TR despite optimal medical therapy (OMT), for whom tricuspid valve replacement is deemed appropriate by a heart team.
The EVOQUE system is comprised of a nitinol self-expanding frame, intra-annular sealing skirt and tissue leaflets made from the company’s proven bovine pericardial tissue. The EVOQUE valve will be available in three sizes, all delivered through the same low-profile transfemoral 28F system.
Successful six-month results from the randomized controlled pivotal trial, TRISCEND II, were presented at TCT 2023 and reported favorable safety and effectiveness outcomes, demonstrating superiority to OMT alone and meeting all primary endpoints. Key findings in the trial included significant reduction or elimination of tricuspid regurgitation and significant and sustained quality of life improvement, while demonstrating a favorable balance between risk and benefit.
In addition to the six-month cohort, 318 of the total 392 randomized patients completed a 1-year visit. The results showed favorable trends in the device group compared to the control group in the primary composite endpoints, including all-cause mortality, tricuspid intervention, heart failure hospitalization, KCCQ, NYHA and 6MWD. Edwards expects to present the full cohort of 392 TRISCEND II pivotal trial patients at TCT 2024.
The EVOQUE system received CE Mark approval in October 2023, making it the world’s first transcatheter valve replacement therapy to receive regulatory approval to treat TR.






















