Over the past ten years, there has been an explosion in neuroendovascular medical device innovation, particularly in the field of acute ischemic stroke. Innovations and improvements in medical device designs have opened the door to acute endovascular treatment, becoming standard of care for large vessel occlusion (LVO), and will likely pave the way for broader indications for ischemic stroke treatment. It is a much better time to be a stroke doctor or a patient now than it was 10 years ago.
First, a little background on the challenges we have faced in LVO treatment.
In 1982, the first reported case of recanalization (i.e. reopening) of a blood vessel occlusion in the brain was reported. This was performed by advancing a microcatheter to the basilar artery in the back of the brain and slowly dripping a thrombolytic agent. This was the first stroke intervention and, for about 20 years, nothing really changed. All a neurointerventionalist could do to treat stroke was get a microcatheter to the clot, drip a thrombolytic over a couple hours, and hope for the best.
In 2001, things started to change. The frustration associated with these long and often unsuccessful procedures led a neurointerventionalist and his colleagues to invent a new corkscrew device called the MERCI retriever, which could be delivered to the clot through a microcatheter, grab the clot, and pull it out. This worked fairly well and led to much better patient outcomes than dripping lytics alone.
The MERCI retriever was studied extensively over the following decade and was the feature device in multiple randomized controlled trials studying the efficacy of thrombectomy for LVO in the late 2000s and early 2010s. Unfortunately, the results of these trials, published in 2013, were negative and the field of neurointervention reached a period of despair as it related to recanalization for acute ischemic stroke.
The device that transformed the stroke space was the stent retriever. Around 2008 , an intrepid neurointerventionalist was in a bind…a middle cerebral artery was occluded. A microcatheter was advanced to the site of the occlusion and thrombolytic was administered, but did nothing. So, the neurointerventionalist partially deployed a stent into the clot to disrupt it, left it up for 20 minutes, and pulled it out. The vessel opened, the patient got better, and voila: the stent retriever was born. Eventually the concept of a temporary stent on a wire took off and it was the widespread adoption of this technique in the early to mid 2010s that led to the publication of multiple positive RCTs in 2015 and beyond that established mechanical thrombectomy as first line treatment of LVO. Over the past decade, we’ve seen an evolution in stent-retriever technology as the devices are now easier to deliver, have a smaller delivery profile, and can even be tailored to have higher or lower radial forces. These devices are still preferred by around 50 percent of neurointerventionalists. I think it is safe to say that these devices aren’t going anywhere.
The idea of simply aspirating the clot was soon to follow. Early reports of aspiration of an occlusive embolus using 045” and 054” catheters were promising but there were several challenges to these technologies. First, delivering these catheters to the M1 segment (or even into the distal internal carotid artery) was often very cumbersome, as these catheters lacked the flexibility to be delivered to the occlusion sites. Second, the inner diameter of these catheters was much too small to remove large thrombus burden and deliver enough suction power to effectively clean a vessel out.
However, over the past decade, there has been a marked improvement in braid, coil, liner, and coating technologies, resulting in larger, more navigable catheters to treat LVO. Aspiration catheters are becoming more and more popular among neurointerventionalists primarily due to their ease of use and especially following the publication of multiple randomized controlled trials showing that they work just as well as stent retrievers.
But the stroke problem is still not solved. Even with our BEST devices, we are only achieving complete reperfusion in about 60 percent of cases. Challenges associated with recanalization of LVOs include delivering catheters to the site of the occlusion in challenging arches and tortuous anatomy, recalcitrant clots that will not come out no matter what forces you apply or what devices you use, fragmentation and embolization of the clot during the procedure, and the new frontier: distal vessel occlusions that require us to overcome navigating catheters SAFELY through tiny, fragile vessels and taking out clots that are often compacted and tough.
Regarding recalcitrant clots, many companies are working on developing superbore 8Fr catheters for thromboaspiration. Having these in the neurointerventionalist’s armamentarium will likely help us get closer to that 100 percent recanalization rate for distal ICA and M1 occlusions. These catheters will require a technological revolution to overcome challenges associated with large inner diameters, thin walls, and kink resistance, but I have no doubt they will be part of our standard armamentarium. 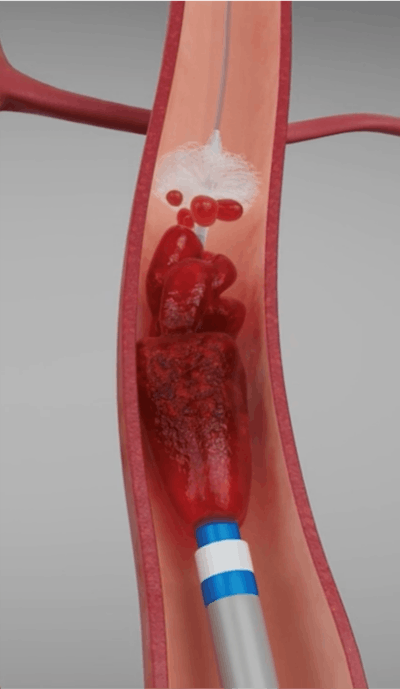 Devices to prevent distal embolization of clots during thrombectomy are on the horizon.MIVI Neuroscience
Devices to prevent distal embolization of clots during thrombectomy are on the horizon.MIVI Neuroscience
One area often overlooked is distal vessel occlusions. Representing nearly 40 percent of symptomatic ischemic stroke cases identified on CTA, such occlusions represent the last frontier of mechanical thrombectomy. Navigation of flexible and kink-resistant thromboaspiration catheters to distal branches, and delivering adequate aspiration power, has proved challenging. However, technological advancements, including development of partial catheters that take advantage of larger proximal IDs, cyclical aspiration technology, smaller stent-retriever designs, and smarter tubing systems may prove to be the trick.
In addition to treating distal emboli, prevention of distal emboli, which can occur in up to 40 percent of cases (as judged by TICI2b rates), may also be a noteworthy innovation. Currently, the only technology available to reduce embolization of clots during the retrieval process are proximal balloon guide catheters, which temporarily arrest anterograde blood flow. But the development of distal protection devices, which are safe for the cerebral vasculature, could help propel the field closer to 100 percent rates of complete revascularization.
In conclusion, while the neurovascular field has made significant advancements since the days of catheter-directed thrombolysis for treatment of ischemic stroke, we still have a long way to go. Rates of complete revascularization are still roughly 60 percent and we still face challenges in catheter deliverability, complete clot ingestion, and navigation and treatment of distal vessel occlusions. Let’s see what the next 10 years brings.
Dr. Waleed Brinjikji is medical director at MIVI Neuroscience, which develops and commercializes devices for complex neurointerventional procedures, and an internationally respected interventional neuroradiologist in Rochester, Minn. Dr. Brinjikji has conducted extensive stroke and neuroendovascular research, with more than 500 peer-reviewed papers and multiple industry and NIH grants, including for a study on improving revascularization techniques in stroke. He trained at top North American institutions, including the University of Toronto. At MIVI, he provides input and medical guidance for clinical research and studies, product design and development, and clinical and customer communications. MIVI’s products are the Q Aspiration Catheter, which optimizes luminal radius and length for optimal aspiration, and the DAISe Thrombectomy Device, designed to reduce distal embolization and improve patient outcomes.























