Neuspera Medical, a medical device company developing implantable devices for patients battling chronic illnesses, said it will begin enrollment in its clinical trial (SANS-UUI), a single-arm study that will enroll 145 patients at 25 sites globally. The study will evaluate the safety and efficacy of Neuspera's Nuvella system designed to treat overactive bladder (OAB) for patients with urinary urgency incontinence (UUI) symptoms.
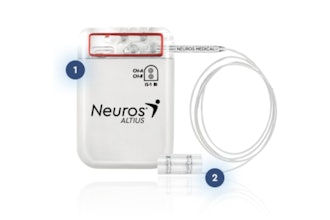
The system is the first sacral neuromodulation (SNM) device that offers a minimally invasive option, which may allow for a better patient experience and greater procedural versatility.
"We are very pleased the SANS-UUI trial has reached this important enrollment milestone, as we are now one step closer to bringing the life-changing benefits of this technology to patients," said Alexander Yeh, Founder and Chief Technology Officer at Neuspera Medical. "The Nuvella system is the smallest available SNM device designed to minimize post-procedure discomfort and have no visible external appearance of the implant."
The feasibility phase results of SANS-UUI were presented at the annual meeting of the American Urological Association (AUA) in September 2021 and at the winter meeting of the Society of Urodynamics, Female Pelvic Medicine & Urogenital Reconstruction (SUFU) in February of this year. In 34 patients implanted with the Nuvella system, 90% of subjects demonstrated a 50% improvement in UUI symptoms at 6 and 12 months with 2 hours of daily stimulation, while 52% were completely dry at the 12-month visit.
"These results are very promising," said Steven Siegel, M.D., Chief Medical Officer at Neuspera Medical. "In addition to the clinical benefits seen, Nuvella demonstrated safety consistent with other commercially available devices."